Capromesh
Partially absorbable
The sterile, partially absorbable, composite mesh is made from two omponents: one absorbable and one non-absorbable.
Capromesh is composed of an absorbable segmented copolymer of glycolide and ε−caprolactone and non-absorbable polypropylene monofilament fibres. The monofilament structure of the mesh decreases the risk of infection and inflammation. After absorption of the absorbable part, only the polypropylene mesh remains in the body. The structure and size of the residual mesh are optimally designed for the physiological stress to which the abdominal wall is subject.
Capromesh is indicated in the operative treatment of fascial defects, in particular hernias, and weakened abdominal wall, the healing of which requires long-term bridging or strengthening of the damaged structures.
Technical information
Fibre thickness:
Approx. 500 μm
Pore size:
2 – 4 mm
Weight:
Initial: approx. 85 g/㎡. PP – contents approx. 28 g/㎡
Stiffness:
Initial: approx. 20 mg. After absorption: approx. 1 mg
Burst strength of the mesh:
Initial: approx. 300 N. After absorption: approx. 175 N
Absorption time:
PGA/PCL part: approx. 90 – 120 days
Advantages
Advantages for the surgeon:
Low inflammation risk due to monofilament structure
Mesh can be cut into any shape and remains stable
Optimal and safe handling during surgery
Suitable for all operation techniques
Made from proven materials: Monolac monofilament (PGA/PCL) and Chiralen (PP)
Good balance between price and quality
Advantages for the patient:
Lower risk of infection
Minimal foreign body component after partial absorption
Optimal elongation in both directions
Maximal comfort for the patient due to optimal softness
Residual weight of the PP-net is only 25 g/m㎡
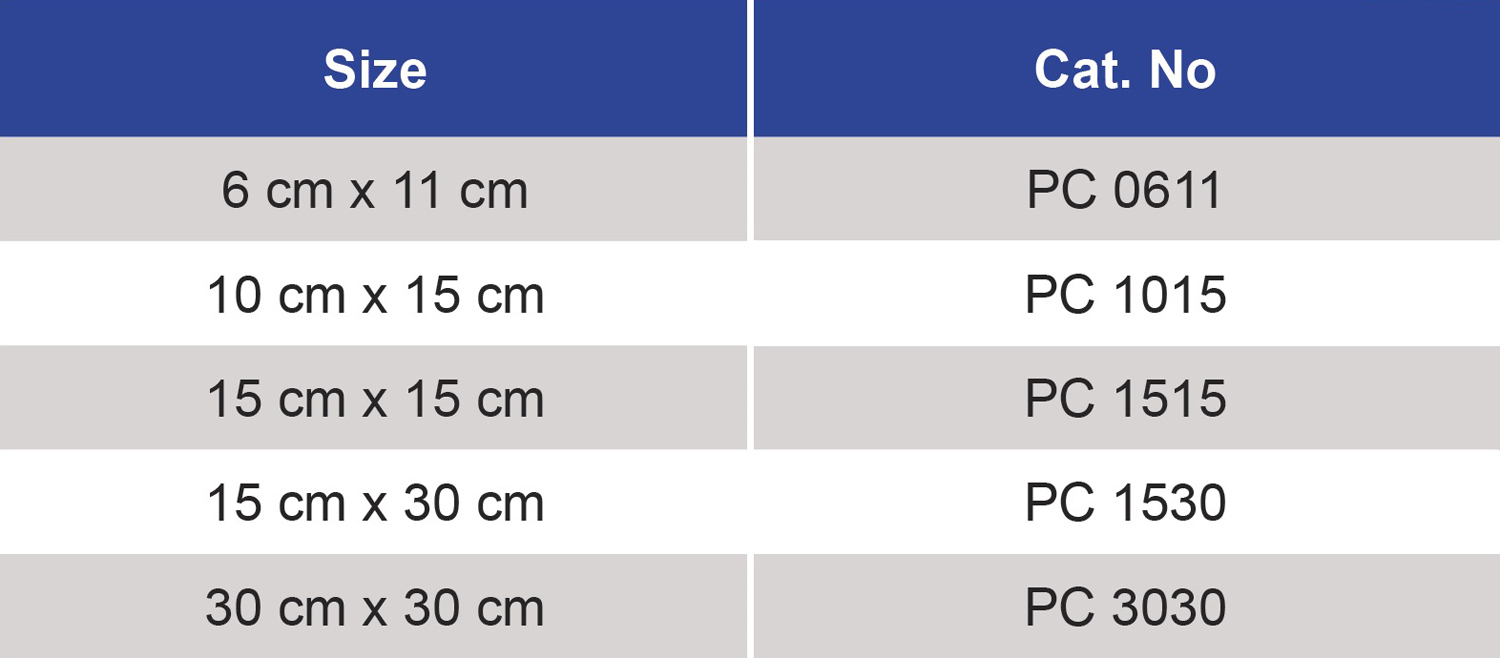
Technical Specifications